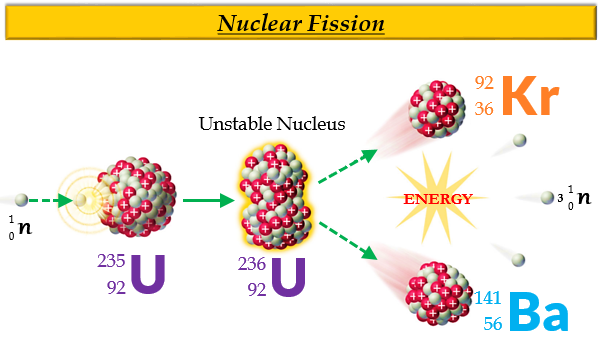
Using the data given, we can write the following initial equation: n 0 1 + P 94 239 u → A 79 204 u + P 15 31 + ? n 0 1 Write the balanced nuclear equation for the process and determine the number of neutrons given off as part of the reaction. Plutonium-239 can absorb a neutron and undergo a fission reaction to produce an atom of gold-204 and an atom of phosphorus-31. Thus, by the careful addition of extra neutrons into a sample of uranium, we can control the fission process and obtain energy that can be used for other purposes. The overall nuclear equation, with energy included as a product, is then as follows: 235U + 1n → 139Ba + 94Kr + 3 1n + energy The reaction can be controlled because the fission of uranium-235 (and a few other isotopes, such as plutonium-239) can be artificially initiated by injecting a neutron into a uranium nucleus. involves the controlled harvesting of energy from fission reactions. Nuclear energy The controlled harvesting of energy from fission reactions. If this energy could be properly harvested, it would be a significant source of energy for our society. Nuclear reactions give off billions of kilojoules per mole. Compare it to combustion reactions of hydrocarbons, which give off about 650 kJ/mol of energy for every CH 2 unit in the hydrocarbon-on the order of hundreds of kilojoules per mole. This is an extraordinary amount of energy. That is, 16.5 billion kJ of energy are given off every time 1 mol of uranium-235 undergoes this nuclear reaction. In the course of the uranium nuclear chemical reaction, the mass difference is converted to energy, which is given off by the reaction: E = (−0.0001834 kg)(3.00 × 10 8 m/s) 2 = −1.65 × 10 13 J = −1.65 × 10 10 kJ 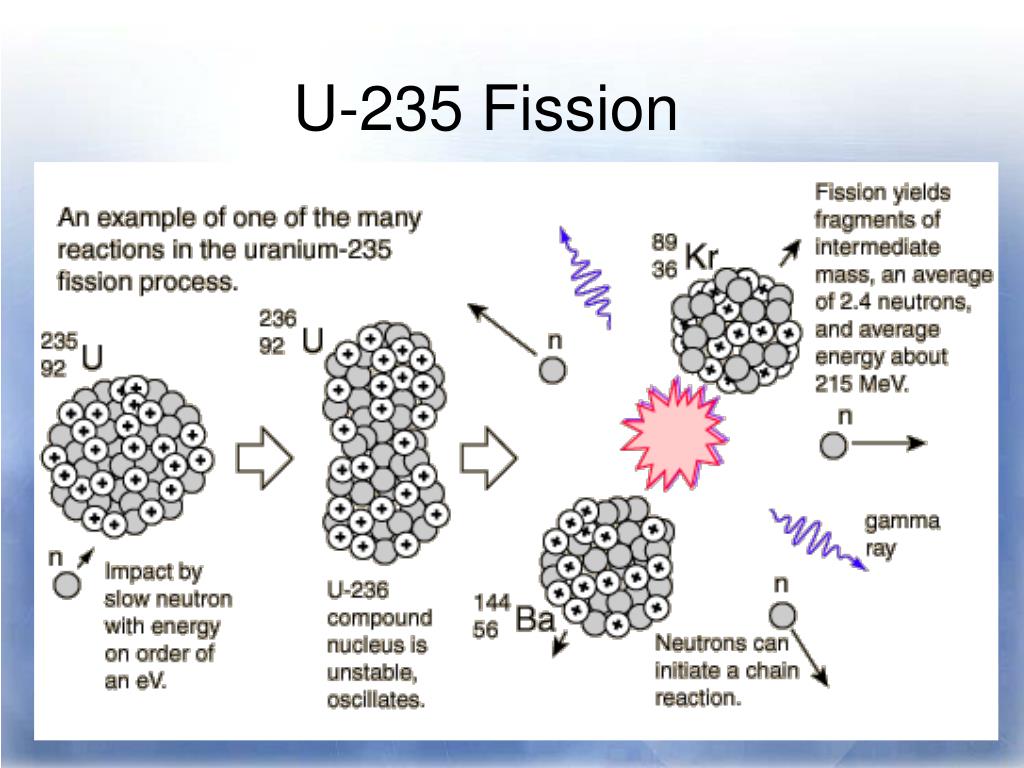
Where c is the speed of light, or 3.00 × 10 8 m/s. Where did this mass go?Īccording to Albert Einstein’s theory of relativity, energy ( E) and mass ( m) are related by the following equation: E = mc 2 If we compare the mass of the reactant (235.0439) to the masses of the products (sum = 234.8605), we notice a mass difference of −0.1834 g, or −0.0001834 kg. Consider the following nuclear reaction, in which the molar mass of each species is indicated to four decimal places: U 235 235.0439 → B 139 a 138.9088 + K 94 r 93.9343 + 2 n 1 2 × 1.0087 Where does this energy come from? If we could precisely measure the masses of the reactants and the products of a nuclear reaction, we would notice that the amount of mass drops slightly in the conversion from reactants to products. Nuclear changes occur with a simultaneous release of energy. Describe the difference between fission and fusion.Explain where nuclear energy comes from.Nuclear fission is a physical process in which the neutron is absorbed in the core of a heavy element and splits it into two lighter cores, converting part of the mass into energy. What is nuclear fission and how does it work? Thus, uranium 235 is the only naturally-occurring element fissile by thermal neutrons all other elements, such as the plutonium, are produced artificially. Before that, it was not known that a neutron could break a nucleus to two parts and release a very large amount of energy. Its main feature is that it is fissile with neutrons, which was accidentally discovered in 1938. It is considered one of the heaviest naturally-occurring elements as it is 18.7 times as dense as water. Uranium was discovered in 1789 by the German chemist Martin Klaproth and it was named after Uranus, the planet, which was discovered a few years earlier. What are the properties of the uranium atom? The most represented isotopes of uranium in nature are uranium 238 (99.29%) and uranium 235 (0.71%). The decay time of uranium is comparable to that of the earth, so it is still found in the Earth's crust. The released energy is stored in the Earth's crust and it contributes to the geothermal energy of our planet. Uranium is not a stable element and it gradually decays, which means that it emits energy. The most economic minerals are uraninite and carnotite and in low concentrations it can even be found in the sea. It can be found in an average concentration of 2 grams per tonne and in various mineral forms. 
Uranium is a chemical element to be found in the Earth's crust ever since the formation of our planet.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
